
The distance between the radii is 266 p m. Beneath the molecule is the label, “B r radius equals 228 p m divided by 2 equals 114 pm.” The fourth diatomic molecule is in purple. The distance between the radii is 228 p m. Beneath the molecule is the label, “C l radius equals 198 p m divided by 2 equals 99 pm.” The third diatomic molecule is in red. The distance between the radii is 198 p m. The second diatomic molecule is in a darker shade of green. These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). Beneath the molecule is the label, “F radius equals 128 p m divided by 2 equals 64 p m.” The next three models are similarly used to show the atomic radii of additional atoms. The periodic table is arranged by atomic weight and valence electrons. The distance between the centers of the two atoms is indicated above the diagram with a double headed arrow labeled, “128 p m.” The endpoints of this arrow connect to line segments that extend to the atomic radii below. Two spheres are pushed very tightly together. The first model, in light green, is used to find the F atom radius. In figure a, 4 diatomic molecules are shown to illustrate the method of determining the atomic radius of an atom. The general trend is that radii increase down a group and decrease across a period. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. Iron has an electron configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6. The number of valence electrons is 3, but they include electrons in a p-type orbital, so to calculate the group number we need to add 10 to the number of valence electrons. the pair of dots represent valence electrons that are not involved in bonding, called lone pair of electrons.\): (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. The highest occupied electron shell is 2, so the period number is 2.Each line in these molecules represents a bonding electron pair, and.Carbon, nitrogen, oxygen, and fluorine with 4, 3, 2, and 1 unpaired dot can make 4, 3, 2, and 1 bond, e.g., in the following molecules:, ,, and. A bond is formed by sharing unpaired valence electrons. A bond is represented by a line between the bonded atoms. Counting valence electrons for main group elements Periodic table Chemistry Khan Academy - YouTube. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. Generally, each unpaired dot can make one bond.įor example, a hydrogen atom with one unaired dot can make one bond as in H-H. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. An element period is a horizontal row on the periodic table. Valence electrons are the electrons present in the outermost shell of an atom. That is, he organized the elements in groups according to their chemical characteristics. Atoms in a group share the same number of valence electrons. A full valence shell is the most stable electron configuration. 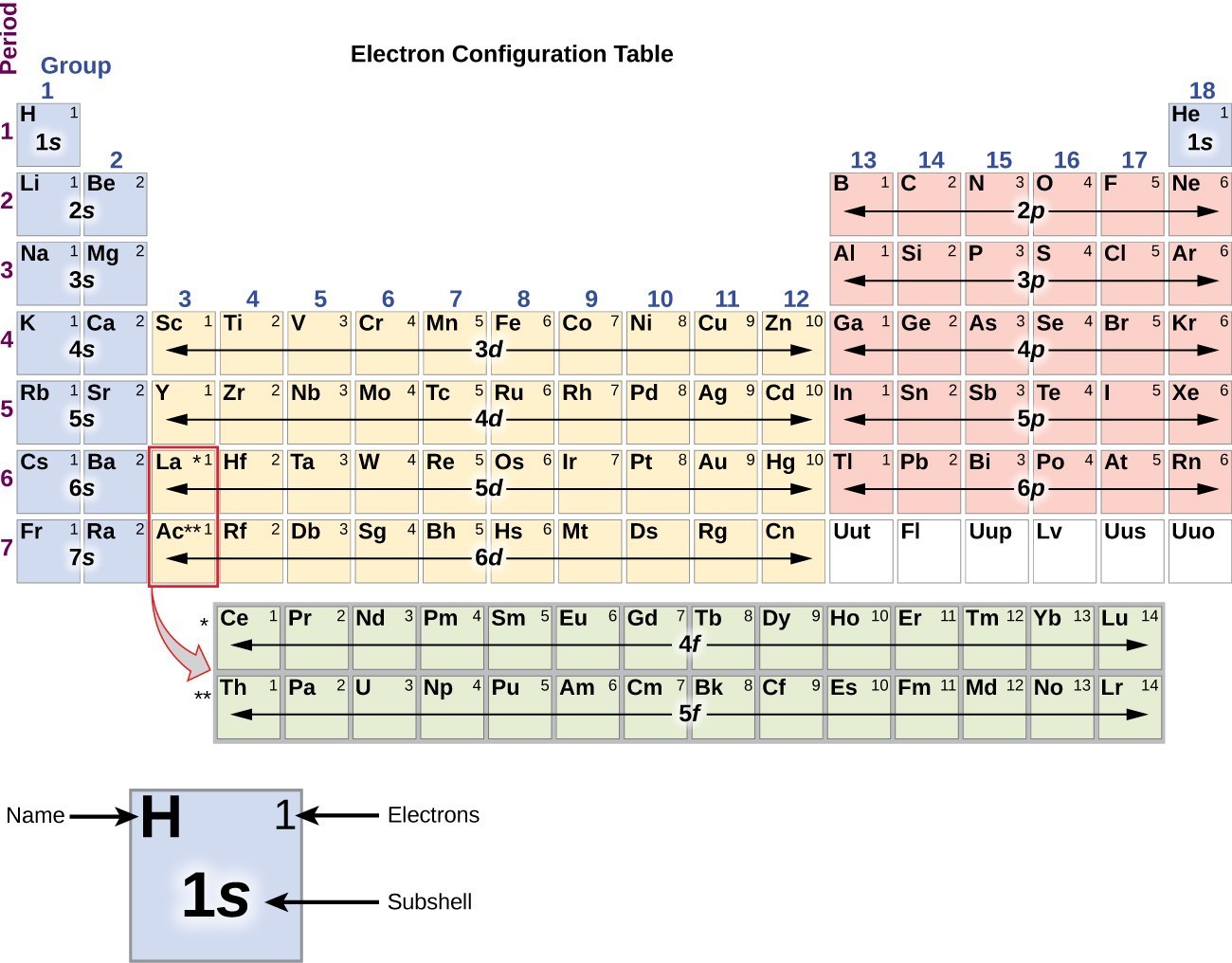
Group 18 elements (helium, neon, and argon are shown in Figure 2) have a full outer, or valence, shell. 
Mendeleev combined the order of atomic weight with common valences to organize the elements in a table. Bohr diagrams indicate how many electrons fill each principal shell. The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make. The valence, in Mendeleevs understanding, was an indication of the ability of an element to combine with other elements. \): Lewis symbols or electron-dot symbols of the first twenty elements in the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
